Biomolecular Systems and Design Lab
We investigate how peptides self-assemble and interact with interfaces to control biological function and dysfunction. We connect mechanisms of peptide aggregation and membrane activity with the rational design of biomolecular materials.
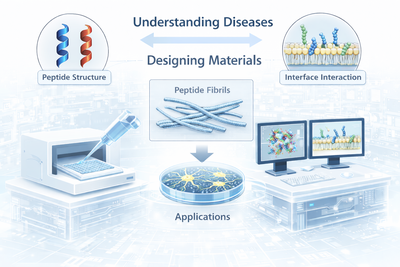
Research Areas
We study how biological and synthetic interfaces, including nanoparticles, surfaces, and soft matter, direct and modulate peptide self-assembly pathways, with a focus on amyloid fibril formation, corona effects, and interface-driven aggregation relevant to neurodegenerative diseases.
We decode how peptide sequence encodes fibril structure, polymorphism, and function. Using molecular simulations and biophysical experiments, we establish sequence–structure–function relationships that guide the rational design of peptide fibrils and hybrid biomaterials for applications in catalysis, regenerative medicine, and biosensing.
We investigate how self-assembling and antimicrobial peptides interact with lipid membranes, exploring how membrane composition, oxidation state, and peptide aggregation govern membrane activity, selectivity, and biological function.
Recent Publications
Join the Lab
We welcome motivated students and researchers with backgrounds in chemistry, biochemistry, biophysics, or computational science. Projects combine experimental and computational approaches.